Liquid-liquid dispersing
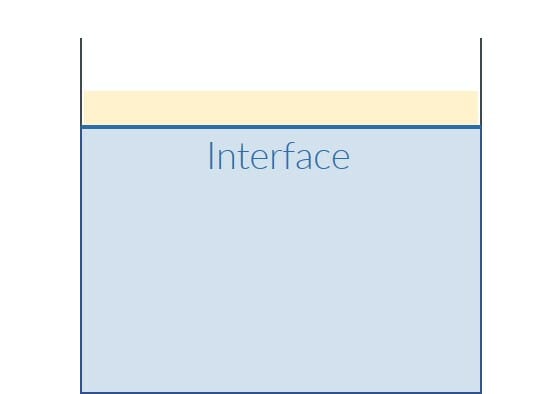
Liquid-liquid dispersing is the process of producing a dispersion. A dispersion is a finely dispersed mixture of at least two normally immiscible liquids. A good example of an everyday dispersion is the long-term stable emulsion, an oil-water mixture. If you pour oil into water, the oil first floats on the surface of the water without mixing with the water. The oil is the so-called inner phase, the water the outer phase.
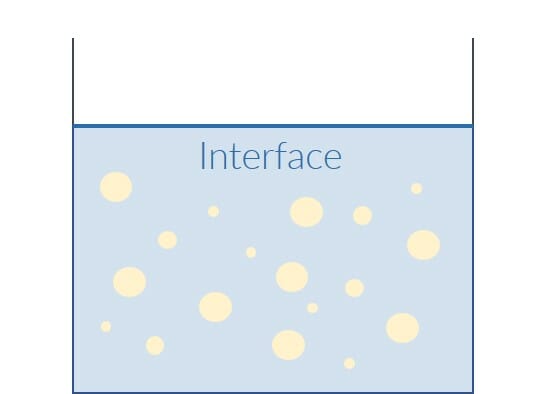
In order to produce a dispersion, high shear forces are used for dispersing. High shear forces are achieved through the use of an impeller with appropriate properties, such as the dissolver disk, and high speeds. So-called emulsifiers are used to stabilize the temporary dispersion. Emulsifiers combine the two substances at the interface because, in our example of the oil-water emulsion, they can bind to both water and oil. This turns the dispersion into a long-term stable emulsion.
Even an emulsion remains unstable, which means that it dissolves into its components again after a relatively short time.

PTM Wiki
There is even more worth knowing in our PTM Wiki.



