Gas-liquid dispersing
Gas-liquid dispersing means the breaking up of gas bubbles in a liquid, also called gassing. This is intended to create the largest possible mass transfer area between the gas and the liquid. In simple terms, this means that the gas should be distributed in the liquid in such a way that as much liquid as possible comes into contact with the gas. The opposite would be the case, for example, if the gas were only applied to the surface of the liquid. Consequently, only the surface of the liquid would be in contact with the gas and only the surface would react with the gas.
The contact between gas and liquid should then result in a reaction. These reactions are required in biotechnology, as well as in the chemical and pharmaceutical industries. Examples of this are hydrogenation (adding hydrogen), oxidation (adding oxygen) and fermentation (aerobic: adding oxygen). The exchange of one gas present in the liquid by another can also be the aim of the dispersing process.
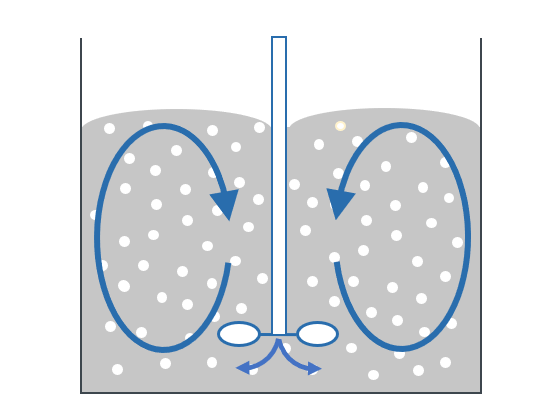
With an optimal process, the gas is broken up into as many small gas bubbles as possible. These are then distributed as evenly as possible in the liquid without creating other counterproductive processes such as foam formation. The gas can be introduced via the surface, via a hollow shaft (see diagram) or via an additional line with pressure.
PTM Wiki
There is even more worth knowing in our PTM Wiki.
Please contact us:
If you would like to be advised on our products, our experts will gladly take care of your request:



