Suspension is the process of dissolving solids in a liquid. The result, i.e. the liquid with finely dispersed solids, is called a suspension. If a suspension is left to stand, the components separate again, the solids with a greater density than the liquid sink back to the bottom and form a sediment.
Initial phase
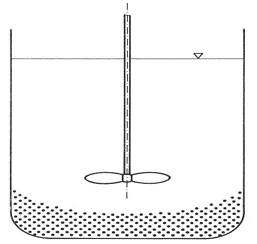
The solids are located at the bottom of the vessel as sediment. They are stirred up according to the one-second criterion. Alternatively, the solids are dosed on top of the surface and then introduced.
Final phase
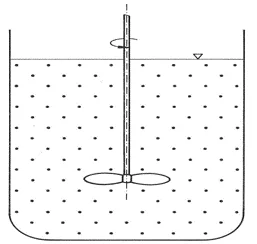
Suspension is complete when a homogeneous distribution of solids has been achieved, i.e. the solids are evenly distributed in the liquid.
Examples of a suspension are mortar or concrete, paint, blood, wheat beer, orange juice or liquid medication with the addition of a drug in powder form.




